 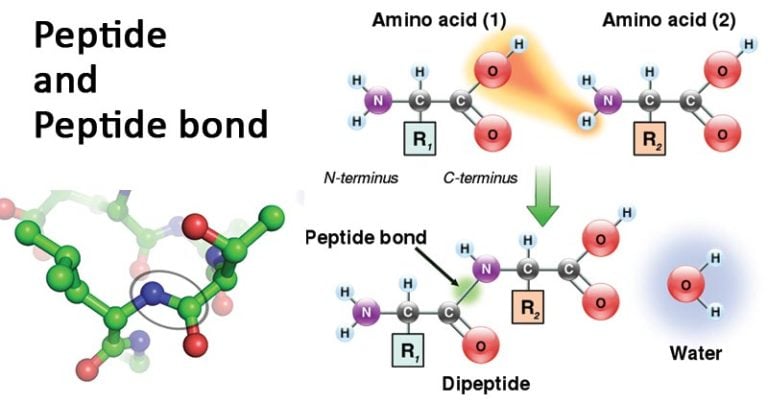
For simplicity, only one kind of side chain is represented (CH 3 for alanine).įig. The backbone chain is formed out of white pieces, while side chains (R groups) are formed out of gray pieces. Rather, it uses color to code for different parts of the polypeptide. The color code used in the pushfit model does not differentiate between different types of atoms. H atomic positions are represented as the ends of short rods coming from the other atoms. The scale for the pushfit models is 1 cm = 1 Å.C, N and O atoms are represented by 0.5 cm diameter balls. We will use a ball and stick modeling system called "pushfit". The atoms can therefore be positioned accurately to correspond to atomic coordinates calculated from geometric considerations or deduced from x-ray crystallographic analysis.When a model has been constructed, interatomic distance can easily be measured. The animation above shows the steps in forming a dipeptide using CPK models.Īlthough space filling models illustrate molecular packing very well, skeletal ("ball and stick") models are easier to work with for most other applications.Their principal advantage is that the positions of the atoms and bonds can easily be seen. Formation of a dipeptide using a CPK model The keyed linker fits into slots in the two atoms.įig. In order to prevent free rotation in the CPK models, a special keyed gray linker is used to connect the nitrogen and carbons of adjacent amino acids. 8 Using the keyed linker in a peptide bond. This is significant because the non-rotatibility of the peptide bond ensures that the oxygen and hydrogens are always oriented in opposite directions relative to the peptide bond.įig. Thus the atoms in the vicinity of the bond (the carbon and nitrogens forming the bond, the oxygen connected to the carbon, and the hydrogen connected to the nitrogen) have fixed positions in the plane that is formed by the bonds between them (rectangle on the right of Fig. 7), the peptide bond has partial double bond character that prevents free rotation around the bond. Sharing of electons among adjacent p orbitals creates resonance structures that give partial double bond character to the peptide bond.īecause of the delocalization of electons from the double-bonded oxygen to the peptide bond (Fig. Image by Alejandro Proto CC BY-SA 3.0 from Wikimedia Commons modified by text translation and croppingįig. The resulting bond between the amino acids is called a peptide bond. 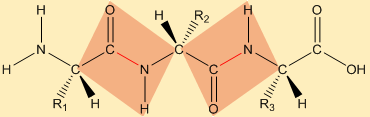
Two amino acids can combine in a condensation reaction (where a molecule of water is formed). Experimental Design Project Toggle Dropdown.11 Reporting the Results of a Statistical Test.6 Scatter plot, trendline, and linear regression.5.6 Discussing statistics in your scientific writing.5.4 A test for differences of sample means: 95% Confidence Intervals.5.2 P and Detecting Differences in Variable Quantities.3.3 Calculating Descriptive Statistics using Excel.3.1 Describing Quantities and Their Variation.Excel Reference and Statistics Manual Toggle Dropdown. 
Appendix A: Calculation Final Concentrations.3.1 Specific details regarding scientific writing.Scientific Literature Guide Toggle Dropdown. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
